The Quiet Burn – What Is Inflammaging, Really?
Most of us are familiar with the obvious signs of inflammation; the redness around a cut; the swelling after a sprain; the heat of a sore throat. These are acute responses; visible, often painful, and temporary. But beneath the surface, a far subtler version can unfold over time – one that rarely announces itself loudly, yet drives the body’s gradual decline. Scientists call it inflammaging; a term coined to capture how chronic, low-grade inflammation accelerates biological ageing (1).
According to recent research, ageing is not a passive march towards decay; it is a progressive accumulation of cellular damage that begins much earlier in life than most of us realise. Central to this process is oxidative stress; damage to mitochondria; the breakdown of DNA repair mechanisms; and the accumulation of senescent cells. These are cells that have stopped dividing, no longer contribute to tissue renewal, and crucially, do not quietly fade away. Instead, they adopt what is known as a senescence-associated secretory phenotype, or SASP. This means they begin secreting inflammatory molecules such as interleukin-6 (IL‑6) and tumour necrosis factor alpha (TNF‑α), which keep nearby tissues in a constant, simmering state of immune activity (2).
Unlike acute inflammation, which peaks and resolves, this chronic version lingers. It does not feel like a fever or look like swelling; rather, it’s a low-level biological ‘hum’ that steadily erodes the body’s repair systems. Over time, this internal inflammation contributes to structural decline across multiple organs, including the skin. Collagen breaks down more easily; repair mechanisms falter; the skin’s barrier weakens; and its defences against environmental stressors become compromised (1,2).
This is not some obscure biological footnote; it plays out in very real, everyday experiences that many simply accept as part of getting older. The skin may look duller or feel drier; elasticity begins to fade; wrinkles deepen and sagging becomes more noticeable. Recovery after sun exposure may take longer; minor irritations linger. You might feel more easily run down when your immune system is under pressure – not quite ill, but not quite right either. These aren’t just superficial changes; they are signs of the body’s internal equilibrium being gradually unsettled (1,2).
Encouragingly, the same research reminds us that ageing is not fixed. The rate at which these molecular processes unfold (particularly the inflammatory ones) is shaped significantly by our choices. Diet; movement; sleep quality; exposure to pollution or chronic stress; all play a role in either accelerating or slowing this trajectory. Inflammaging is not inevitable; it is, at least to a degree, modifiable (1,2).
And increasingly, the conversation is turning to one organ in particular as a powerful lever for change: the gut (2).
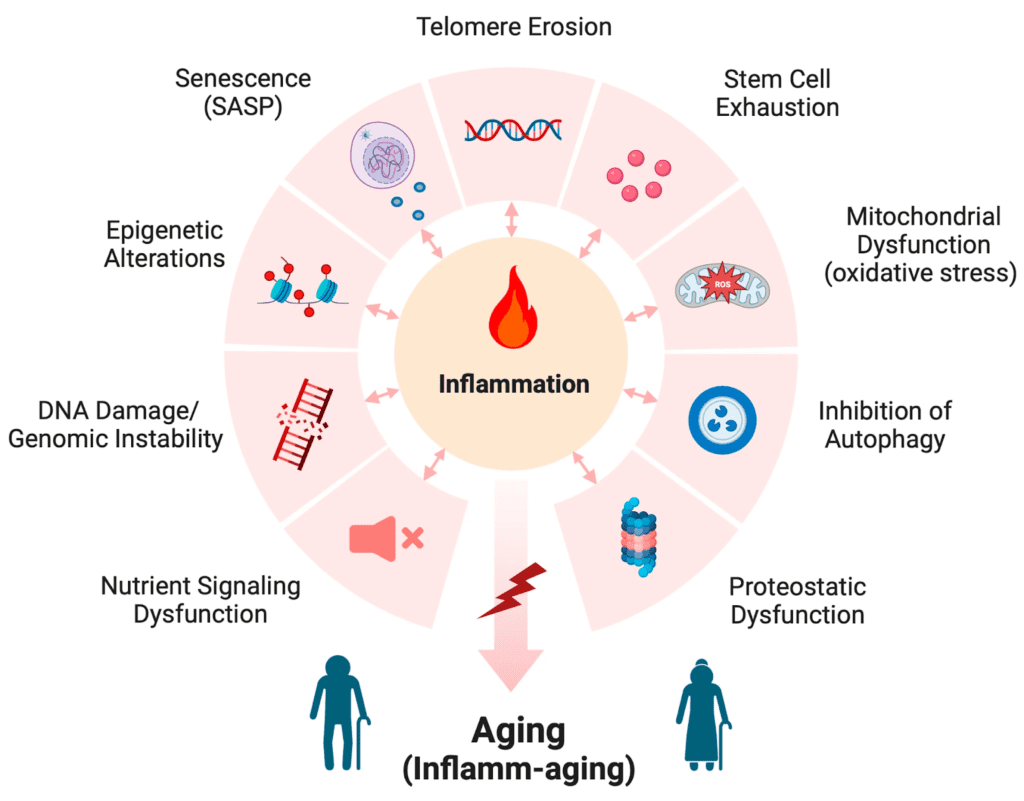
Agrawal, R., Hu, A. and Bollag, W.B. (2023). The Skin and Inflamm-Aging. Biology, 12(11), p.1396. (3)
A visual summary of the cellular processes driving inflammaging; chronic inflammation fuels key hallmarks of ageing such as mitochondrial dysfunction, DNA damage, senescent cell accumulation (SASP), and impaired autophagy – all contributing to systemic decline over time.
Why We Feel Older Than We Are; The Hidden Toll of Low-Grade Inflammation
We often talk about ageing as though it begins with visible signs – wrinkles, stiffness, or grey hair – but in truth, it starts long before any of those surface changes appear. One of the most overlooked players in this process is low-grade inflammation; not the dramatic swelling of an injury, but something far more insidious. It’s a kind of background biological noise; quiet, persistent, and deeply energy-draining. And although it rarely causes immediate alarm, its long-term impact can shape how old we feel – and eventually, how long and well we live (4).
This subtle form of immune activation doesn’t announce itself with dramatic symptoms. Instead, it wears us down slowly. People often describe feeling more tired than usual; struggling with brain fog; noticing poorer sleep or a dip in mood they can’t quite explain. Muscles may feel weaker than they used to; joints stiffer in the morning; the skin perhaps a little duller or slower to heal. These are the moments that many chalk up to “just getting older” – and yet, at the molecular level, there’s more going on (4).
That tired, worn-down feeling often reflects the constant circulation of inflammatory signals in the body. Over time, these signals affect everything from how we produce energy, to how we think and recover. The immune system becomes more reactive, but paradoxically, less efficient; minor infections become harder to shake; exercise recovery slows. The body feels heavier – not just physically, but in terms of sheer effort required to feel well (4).
At the centre of this process are cytokines; tiny signalling molecules such as interleukin-6 (IL‑6), tumour necrosis factor alpha (TNF‑α), and interleukin-1 beta (IL‑1β). These molecules are essential in short-term immune responses, but when their levels remain subtly elevated over months and years, they begin to alter the body’s internal balance. They can interfere with metabolism; change how brain cells communicate; and slowly rewire immune responses. In this prolonged state, they’ve been linked with increased risks of cardiovascular disease, type 2 diabetes, frailty, and cognitive decline. These aren’t theoretical connections; they’re increasingly backed by long-term epidemiological and mechanistic studies (4).
This kind of chronic inflammation also has a damaging companion: oxidative stress. Inflammatory cells produce reactive oxygen species (ROS), which can damage DNA, proteins, and cellular membranes. That damage then triggers more inflammation, creating a vicious cycle where the two processes feed into one another. Oxidative stress fuels inflammation; inflammation fuels more oxidative stress. Over time, this loop accelerates biological ageing in the arteries, the brain, the joints, and even the skin. The impact isn’t just cosmetic; it’s systemic (4).
This is precisely where a platform like SoLongevity finds its footing. Rather than simply treating symptoms downstream – stiffness, tiredness, or visible signs of ageing – its approach focuses on calming inflammation at its source by reducing oxidative stress. This is not about extreme interventions or overblown claims; it’s about creating the conditions in which the body can operate more smoothly, more quietly, and more resiliently. In this framework, the “anti-inflammatory lifestyle” becomes synonymous with a “longevity lifestyle”; one that integrates nutrition, movement, sleep, stress management, and targeted compounds to dial down that background immune noise (4).
Importantly, we’re now seeing more scientific consensus around the link between chronic inflammation and the major diseases of ageing. Conditions like atherosclerosis, insulin resistance, type 2 diabetes, osteoporosis, cognitive decline, and even sarcopenia – the age-related loss of muscle – all share common inflammatory pathways. This doesn’t mean inflammation is the sole cause; ageing is never that simple. But it does mean that ongoing, low-level immune activity is one of the threads running through many of these conditions (4).
What this tells us is that the invisible forces behind everyday fatigue, stiffness, and brain fog are often the same ones that, over decades, shape our risk of chronic illness. They aren’t separate issues; they’re points on the same spectrum. And that means that addressing inflammation today is not just about feeling better now – it’s a strategic investment in our long-term health and capacity (4).
Your Gut: The Anti-Ageing Firewall You Never Knew You Had
For something so central to our health, the gut is often misunderstood. We associate it with digestion, bloating, or the occasional dietary regret. But the gut, and more specifically, the vast microbial ecosystem it hosts, plays a far more profound role in shaping how we age. It is increasingly being recognised as a kind of biological firewall; one that, when intact, keeps systemic inflammation in check; and when compromised, allows inflammaging to spread (2).
As we age, our gut microbiota begins to shift. Microbial diversity tends to decline; populations of beneficial, short-chain fatty acid (SCFA)-producing bacteria such as certain Clostridia, Ruminococcaceae, Akkermansia, and Bifidobacterium often decrease; and more pro-inflammatory strains can take their place. This imbalance, known as dysbiosis, weakens the gut barrier, a thin but vital layer that separates the gut’s contents from the bloodstream (2).
When this barrier is intact, it functions like a gatekeeper; selectively allowing nutrients through while keeping potential threats out. But with dysbiosis, the barrier becomes leaky. Bacterial components such as lipopolysaccharides cross into the bloodstream and activate the immune system. This leads to elevated levels of circulating inflammatory molecules; the same ones involved in inflammaging, including TNF‑α, IL‑1β, and IL‑6 (2).
This constant, low-level exposure to microbial signals has been proposed as a key driver of chronic inflammation. It feeds into cellular senescence and the ageing of the immune system itself, known as immunosenescence. The more compromised the gut becomes, the more it contributes to this persistent inflammatory state; a subtle but significant accelerant to biological ageing (2).
One of the clearest illustrations of this dynamic is what researchers call the gut–skin axis. In a well-functioning system, fibre in the diet is fermented by beneficial gut microbes to produce SCFAs; including butyrate, acetate, and propionate. These molecules support the integrity of the gut lining; reduce inflammation; and create an immune environment that protects and nourishes the skin. Their effects travel through the circulation, supporting hydration, elasticity, and even the skin’s ability to recover from environmental stress (2).
When the gut is disrupted, however, this protective system falters. Pro-inflammatory metabolites and SASP factors begin to circulate more freely. These can activate enzymes in the skin, particularly matrix metalloproteinases, that accelerate the breakdown of collagen and other structural proteins. The result is more visible wrinkling; sagging; dryness; and barrier dysfunction. These are not merely surface-level issues; they are visible reflections of internal imbalance (2,3).
What’s promising is that these age-related changes to the gut and its microbial residents are not beyond our control. The review you provided makes clear that dietary interventions (particularly through prebiotic fibres and targeted probiotics) can support the growth of SCFA-producing bacteria and help restore immune balance. In older adults, such interventions have been shown to improve skin hydration, elasticity, and reduce wrinkle depth, presumably through this same gut–skin inflammatory axis (2,3).
This is where fibre-rich, microbiome-supporting drinks begin to take on a new significance. They are no longer simply about easing digestion; they become part of a broader strategy for managing ageing at the cellular level. By supporting microbial diversity and SCFA production, these drinks help to reinforce the gut barrier; quieten chronic inflammation; and protect downstream systems – including the brain, immune system, and skin (2).
Ratanapokasatit et al. (2022) (2)
Illustrating current perspectives on how gut and skin microbiomes influence the ageing process through inflammatory and barrier-modulating pathways.
Feeding the Firewall – How Fibre Nourishes the Anti-Ageing Microbiome
The link between ageing and inflammation isn’t just theoretical; it’s functional. And one of the most consistently overlooked tools for intervening in this process is dietary fibre. Not the cardboard-tasting kind we associate with digestive remedies, but diverse, colourful, plant-based fibre that feeds the gut microbiome and boosts the production of short-chain fatty acids like butyrate – the same anti-inflammatory compounds that help to seal the gut barrier, regulate immune responses, and protect skin, brain and metabolic health as we age (5).
This is where a thoughtful, well-formulated drink can become far more than a treat or a convenience. It becomes part of a daily nutritional strategy to support longevity from the inside out. At FiiHii, each Frink® has been designed to deliver more than flavour; it provides functional nutrition with a specific purpose. Here’s how each one supports the microbiome and helps push back against inflammaging:
FiiHii Frinks® – Supporting Gut Health, Fibre Diversity and Anti-Inflammaging Nutrition
Cocomangofango Frink
– High in soluble and insoluble fibre from tropical fruits
– Bromelain and vitamin C support anti-inflammatory action
– Coconut fats help absorb fat-soluble antioxidants
– Supports microbiome diversity and collagen production
Orchard Frink
– Rich in pectin, a prebiotic fibre that nourishes gut bacteria
– Antioxidants from pomegranate and citrus support immune health
– Pumpkin seeds add zinc and healthy fats for microbiome support
P-Power Frink
– Clinically-backed fibre from prunes supports bowel regularity
– Omega-3 from flax helps balance inflammation
– Polyphenols aid mood and gut–brain axis function
Traffic Light Punch Frink
– Fibre-rich berries and chia promote microbial diversity
– Omega-3 and polyphenols reduce systemic inflammation
– Supports hydration and gut motility
Summer Love Frink
– A blend of calcium, vitamin C and iron from fruit and seeds
– Antioxidants and fibre enhance skin and gut health
– Promotes metabolic balance and immune resilience
The God Yoghurt Frink
– Avocado and hemp provide essential fats for nutrient absorption
– Spinach and bananas offer fibre and magnesium for stress support
– Blueberries and polyphenols target inflammation and oxidative stress
By combining these ingredients into delicious, easy-to-drink blends, FiiHii Frinks® offer a daily dose of microbiome-supportive fibre, essential nutrients and anti-inflammatory compounds – without feeling like a chore. It bridges the gap between scientific insight and real-world nourishment; helping to transform the idea of healthy ageing from abstract theory into something practical, pleasurable and sustainable..
Start getting things as they should be here – Buy Now!!
References:
- Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505–22. doi:10.1038/s41569-018-0064-2.
- Ratanapokasatit Y, Laisuan W, Rattananukrom T, Petchlorlian A, Thaipisuttikul I, Sompornrattanaphan M. How microbiomes affect skin aging: the updated evidence and current perspectives. Life. 2022;12(7):936. doi:10.3390/life12070936.
- Agrawal R, Hu A, Bollag WB. The skin and inflamm-aging. Biology. 2023;12(11):1396. doi:10.3390/biology12111396.
- Fülöp T, Larbi A, Witkowski JM. Human inflammaging. Gerontology. 2019;65(5):495–504. doi:10.1159/000497375.
- Fu J, Zheng Y, Gao Y, Xu W. Dietary fiber intake and gut microbiota in human health. Microorganisms. 2022;10(12):2507. doi:10.3390/microorganisms10122507.





